114027
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

从奥尔波特综合征 (Alport syndrome) 患者的肾小管细胞中产生诱导性多功能干细胞
Authors Chen WB, Huang JR, Yu XQ, Lin XC, Dai Y
Received 30 March 2015
Accepted for publication 2 June 2015
Published 21 August 2015 Volume 2015:8 Pages 101—109
DOI http://dx.doi.org/10.2147/IJNRD.S85733
Checked for plagiarism Yes
Review by Single-blind
Peer reviewer comments 4
Editor who approved publication: Professor Pravin Singhal
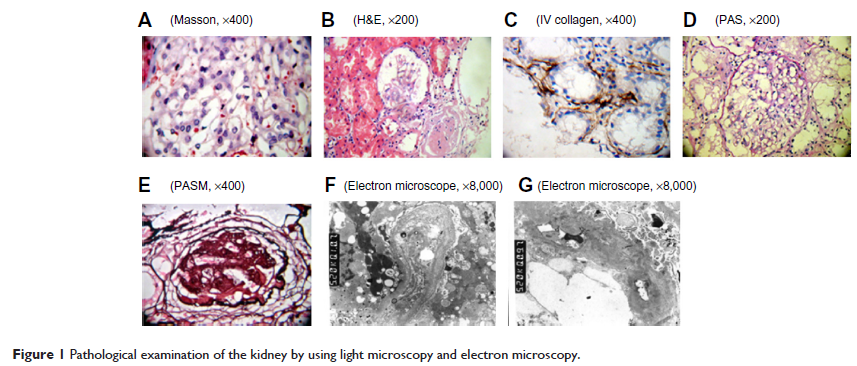
Abstract: Alport syndrome (AS) is a hereditary disease that leads to kidney failure and is caused by mutations in the COL4A3 , COL4A4 , and COL4A5 genes that lead to the absence of collagen α3α4α5 (IV) networks in the mature kidney glomerular basement membrane. Approximately 80% of AS is X-linked because of mutations in COL4A5 , the gene encoding the alpha 5 chain of type IV collagen. To investigate the pathogenesis of AS at the genetic level, we generated induced pluripotent stem cells (iPSCs) from renal tubular cells of a patient with AS. The successful iPSC generation laid the foundation to master the repair of the COL4A5 gene and to evaluate the differentiation of iPSC into Sertoli cells and the accompanying epigenetic changes at each stage. The generation of iPSCs from AS patients not only confirms that iPSCs could be generated from renal tubular cells, but also provides a novel type of genetic therapy for AS patients. In this study, we generated iPSCs from renal tubular cells via ectopic expression of four transcription factors (Oct4 , Sox2 , c-myc , and Klf4 ). According to the human embryonic stem cell (hESC) charter, iPSC formation was confirmed by comparatively analyzing hESC markers via colony morphology, immunohistochemistry, qRT-PCR, flow cytometry, gene expression profiling of the three germ layers, and karyotyping. Our results demonstrated that iPSCs were similar to hESCs with regard to morphology, proliferation, hESC-specific surface marker expression, and differentiation into the cell types of the three germ layers. The efficient generation of iPSCs from the renal tubular cells of an AS patient would provide a novel model to investigate the mechanisms underlying AS and to develop new treatments for AS.
Keywords: Alport syndrome, induced pluripotent stem cells, renal tubular cells