113838
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

将他克莫司与共晶油基微乳液相融合治疗特应性皮炎:同时增强经皮给药和治疗功效,减轻副作用
Authors Wang Y, Cao S, Yu K, Yang F, Yu X, Zhai Y, Wu C, Xu Y
Received 14 April 2019
Accepted for publication 1 July 2019
Published 30 July 2019 Volume 2019:14 Pages 5849—5863
DOI https://doi.org/10.2147/IJN.S212260
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Alicia Fernandez-Fernandez
Peer reviewer comments 2
Editor who approved publication: Dr Lei Yang
Background: Topical application of tacrolimus (FK506) was effective in treating atopic dermatitis (AD); however, the therapeutic efficiency is hampered by its poor penetration into the skin and local side effects of transient irritation symptoms with a burning sensation, a feeling of warmth or heat. Menthol and camphor have been widely used in topical compound formulations for adjunctive pharmacotherapy for antipruritics and analgesics owing to their cool nature, and both present skin penetration enhancing effects. Moreover, they can form a liquid eutectic oil to solubilize hydrophobic drugs.
Purpose: Taking advantages of menthol/camphor eutectic (MCE), this work aims to integrate FK506 into MCE to construct a microemulsion system, i.e., FK506 MCE ME, which simultaneously enhances the percutaneous delivery and treatment efficacy, while reduces the side effects of FK506.
Methods: The formulation of FK506 MCE ME was optimized and characterized. Different formulations containing FK506 were topically administered to treat 1–chloro–2, 4–dinitrobenzene (DNCB)-induced murine AD.
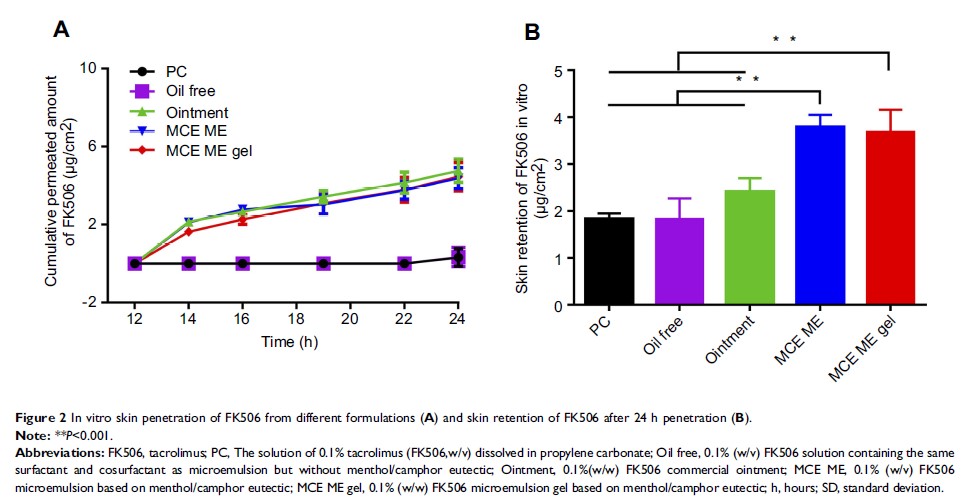
Results: MCE solubilized FK506. FK506 in MCE ME penetrated skin in vitro more than in the commercial ointment, and MCE predominantly exerted the enhancing effects in MCE ME. FK506 MCE ME or FK506 MCE ME gel had greater effects on clinical symptoms, histological analysis, and IgE than did commercial FK506. The anti-pruritic and down-regulation of substance P effects of MCE ME vehicle mitigated the side effects of FK506 application.
Conclusion: MCE ME presented the excellent properties of simultaneously enhancing the percutaneous delivery and treatment efficacy, while reducing the side effects of FK506 for AD. Therefore, MCE ME is a promising nanoscale system for FK506 to effectively treating AD with low irritation and high medication adherence.
Chemical compounds studied in this article: Tacrolimus (PubChem CID: 445643); menthol (PubChem CID: 1254); camphor (PubChem CID: 2537)
Keywords: tacrolimus, microemulsion, menthol, camphor, eutectic, atopic dermatitis, percutaneous delivery