113568
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

新辅助化疗治疗 Her-2 阳性乳腺癌的疗效比较和生存分析
Authors Li S, Wei W, Jiang Y, Li Q, Huang Q, Yang H, Liu J
Received 19 April 2018
Accepted for publication 14 August 2018
Published 21 September 2018 Volume 2018:12 Pages 3085—3093
DOI https://doi.org/10.2147/DDDT.S171534
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Colin Mak
Peer reviewer comments 3
Editor who approved publication: Dr Anastasios Lymperopoulos
Purpose: The objective of this research was to compare the short- and long-term efficacy of the following four neoadjuvant chemotherapy (NAC) regimens: docetaxel/carboplatin/trastuzumab (TCH), docetaxel/epirubicin/cyclophosphamide (TEC), Xeloda/epirubicin/cyclophosphamide followed by Xeloda/docetaxel (XEC-XT), and 5-fluorouracil/epirubicin/cyclophosphamide followed by docetaxel (FEC-T) in human epidermal growth factor receptor-2-positive (Her-2-positive) breast cancer.
Patients and methods: According to treatment preferences, 139 patients with Her-2-positive breast cancer were divided into the following four groups: 39 patients in the TCH group, 35 patients in the TEC group, 33 patients in the XEC-XT group, and 32 patients in the FEC-T group. The primary end points were disease-free survival (DFS) and 5-year overall survival (5-year OS). The secondary end points were the efficacy and toxicity of NAC.
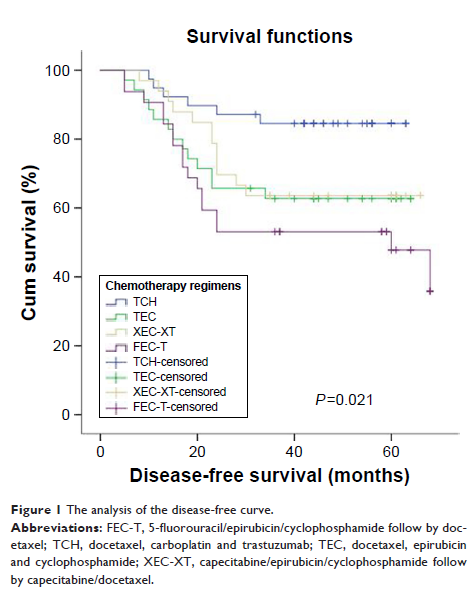
Results: The TCH, TEC, XEC-XT, and FEC-T groups demonstrated overall response rates of 87.1%, 74.3%, 75.8%, and 62.5% (P =0.031), respectively, and pathological complete response rates of 25.6%, 18.2%, 20.0%, and 18.2% (P =0.041), respectively. The DFS rates for the TCH, TEC, XEC-XT, and FEC-T groups were 84.6%, 62.9%, 65.7%, and 46.9% (P =0.01), respectively. The 5-year OS rates for the TCH, TEC, XEC-XT, and FEC-T groups were 87.2%, 69.7%, 71.4%, and 59.4% (P =0.069), respectively. The mean survival time was 59.3 months (TCH group), 53.5 months (TEC group), 55.3 months (XEC-XT group), and 52.4 months (FEC-T group). The difference in survival among the four groups was statistically significant (P =0.04).
Conclusion: In four NAC regimens for the treatment of Her-2-positive breast cancer, the TCH group exhibited better DFS and 5-year OS. The TCH regimen significantly enhanced the pathological complete remission rate of NAC with similar side effects compared to the TEC, XEC-XT, and FEC-T regimens. In terms of long-term efficacy, the XEC-XT treatment was superior to the FEC-T and TEC treatment, and there was no significant difference between the FEC-T and TEC groups.
Keywords: Her-2-positive breast cancer, neoadjuvant chemotherapy, trastuzumab, efficacy, survival analysis, toxic side effects