114027
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

已发表论文
通过改善细胞内给药,使聚乙二醇-聚己酸内酯嵌段共聚物和基于磷脂的隐形纳米颗粒对患有乳腺癌的小鼠具有增强的疗效
Authors He XD, Li L, Su H, Zhou DL, Song HM, Wang L, Jiang XH
Published Date March 2015 Volume 2015:10 Pages 1791—1804
DOI http://dx.doi.org/10.2147/IJN.S75186
Received 30 September 2014, Accepted 13 December 2014, Published 5 March 2015
Background: Effective anticancer drug delivery to the tumor site without rapid body
clearance is a prerequisite for successful chemotherapy.
1,2-distearoyl-sn-glycero-3-phospho-ethanolamine-N-(methoxy[polyethyleneglycol]-2000)
(DSPE-PEG2000) has been widely used in the preparation of stealth liposomes.
Although PEG chains can efficiently preserve liposomes from rapid clearance by
the reticuloendothelial system (RES), its application has been hindered by poor
cellular uptake and unsatisfactory therapeutic effect.
Methods: To address the dilemma, we presented a facile approach to fabricate novel stealth nanoparticles generated by poly(ethylene glycol)-block-poly(ε-caprolactone) (PEG-b -PCL), soybean phosphatidylcholine (SPC), and cholesterol, namely LPPs (L represented lipid and PP represented PEG-b -PCL), for the delivery of anticancer drug paclitaxel (PTX). LPPs were prepared using the thin film hydration method. Two PEG-b -PCL polymers with different molecular weights (MW; PEG2000-b-PCL2000, MW: 4,000 Da and PEG5000-b-PCL5000, MW: 10,000 Da) were used to fabricate stealth nanoparticles. Conventional PEGylated liposome (LDP2000, L represented lipid and DP2000 represented DSPE-PEG2000) composed of SPC, cholesterol, and DSPE-PEG2000 was used as the control. The physical properties, cellular uptake, endocytosis pathway, cytotoxicity, pharmacokinetics, tumor accumulation, and anticancer efficacy of free PTX, PTX-loaded LPPs, and LDP2000 were systemically investigated after injection into 4T1 breast tumor–bearing mice.
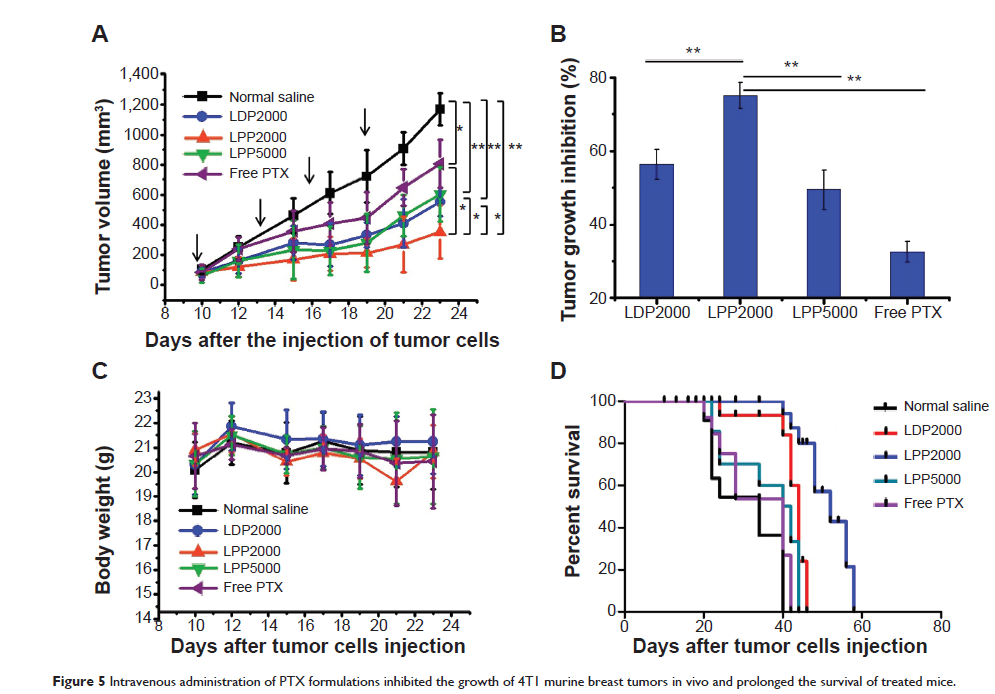
Results: LPPs were vesicles around 100 nm in size with negative zeta potential. With enhanced stability, LPPs achieved sustainable release of cancer therapeutics. The cellular uptake level was closely related to the PEG chain length of PEG-b -PCL; a shorter PEG chain resulted in higher cellular uptake. Moreover, the cellular internalization of LPP2000 modified by PEG2000-b -PCL2000 on 4T1 cells was 2.1-fold higher than LDP2000 due to the improved stability of LPP2000. The cytotoxicity of PTX-loaded LPP2000 was also higher than that of LDP2000 and LPP5000 as observed using a WST-8 assay, while blank LPPs showed negligible toxicity. Consistent with the results of the in vitro study, in vivo experiments showed that LPPs allowed significantly improved bioavailability and prolonged T1/2ß as compared to free PTX injection. More importantly, LPPs mainly accumulated at the tumor site, probably due to the enhanced permeation and retention effect (EPR effect). As a nanomedicine, LPP2000 (tumor inhibition rate of 75.1%) significantly enhanced the therapeutic effect of PTX in 4T1 breast tumor–bearing mice by inhibiting tumor growth compared to LDP2000 and LPP5000 (tumor inhibition rates of 56.3% and 49.5%, respectively).
Conclusion: Modification of liposomes with PEG2000-b -PCL2000 can simultaneously improve drug accumulation at the target tumor site and tumor cells, showing great promise for utilization as a PEG modification tool in the fabrication of stealth nanoparticles for cancer chemotherapy.
Keywords: nanoparticles PEG-b-PCL, phospholipid, murine breast cancer chemotherapy, paclitaxel
Methods: To address the dilemma, we presented a facile approach to fabricate novel stealth nanoparticles generated by poly(ethylene glycol)-block-poly(ε-caprolactone) (PEG-b -PCL), soybean phosphatidylcholine (SPC), and cholesterol, namely LPPs (L represented lipid and PP represented PEG-b -PCL), for the delivery of anticancer drug paclitaxel (PTX). LPPs were prepared using the thin film hydration method. Two PEG-b -PCL polymers with different molecular weights (MW; PEG2000-b-PCL2000, MW: 4,000 Da and PEG5000-b-PCL5000, MW: 10,000 Da) were used to fabricate stealth nanoparticles. Conventional PEGylated liposome (LDP2000, L represented lipid and DP2000 represented DSPE-PEG2000) composed of SPC, cholesterol, and DSPE-PEG2000 was used as the control. The physical properties, cellular uptake, endocytosis pathway, cytotoxicity, pharmacokinetics, tumor accumulation, and anticancer efficacy of free PTX, PTX-loaded LPPs, and LDP2000 were systemically investigated after injection into 4T1 breast tumor–bearing mice.
Results: LPPs were vesicles around 100 nm in size with negative zeta potential. With enhanced stability, LPPs achieved sustainable release of cancer therapeutics. The cellular uptake level was closely related to the PEG chain length of PEG-b -PCL; a shorter PEG chain resulted in higher cellular uptake. Moreover, the cellular internalization of LPP2000 modified by PEG2000-b -PCL2000 on 4T1 cells was 2.1-fold higher than LDP2000 due to the improved stability of LPP2000. The cytotoxicity of PTX-loaded LPP2000 was also higher than that of LDP2000 and LPP5000 as observed using a WST-8 assay, while blank LPPs showed negligible toxicity. Consistent with the results of the in vitro study, in vivo experiments showed that LPPs allowed significantly improved bioavailability and prolonged T1/2ß as compared to free PTX injection. More importantly, LPPs mainly accumulated at the tumor site, probably due to the enhanced permeation and retention effect (EPR effect). As a nanomedicine, LPP2000 (tumor inhibition rate of 75.1%) significantly enhanced the therapeutic effect of PTX in 4T1 breast tumor–bearing mice by inhibiting tumor growth compared to LDP2000 and LPP5000 (tumor inhibition rates of 56.3% and 49.5%, respectively).
Conclusion: Modification of liposomes with PEG2000-b -PCL2000 can simultaneously improve drug accumulation at the target tumor site and tumor cells, showing great promise for utilization as a PEG modification tool in the fabrication of stealth nanoparticles for cancer chemotherapy.
Keywords: nanoparticles PEG-b-PCL, phospholipid, murine breast cancer chemotherapy, paclitaxel