112158
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

中药黄芪多糖增强胰腺癌细胞血管生成抑制剂阿帕替尼的抗肿瘤作用,包括抑制增殖、侵袭和凋亡作用
Authors Wu J, Wang J, Su Q, Ding W, Li T, Yu J, Cao B
Received 15 November 2017
Accepted for publication 20 March 2018
Published 9 May 2018 Volume 2018:11 Pages 2685—2698
DOI https://doi.org/10.2147/OTT.S157129
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Justinn Cochran
Peer reviewer comments 3
Editor who approved publication: Dr Ingrid Espinoza
Background: Traditional chemotherapy and molecular targeted therapy have shown
modest effects on the survival of patients with pancreatic cancer. The current
study aimed to investigate the antitumor effects of apatinib, Astragalus polysaccharide
(APS), and the combination of both the drugs in pancreatic cancer cells and
further explore the molecular mechanisms in vitro.
Materials and
methods: Expression of vascular endothelial
growth factor receptor-2 (VEGFR-2) in human pancreatic cancer cell lines
ASPC-1, PANC-1, and SW1990 was detected by Western blotting. Cell proliferation
was measured by MTS, and migration and invasion were detected by wound-healing
and Transwell assays, respectively. Cell apoptosis rate was determined by flow
cytometry and cellular autophagy level affected by apatinib, and APS was
analyzed by Western blotting.
Results: Human pancreatic cancer cell lines ASPC-1 and PANC-1 expressed
VEGFR-2, but VEGFR-2 was not detected in SW1990. Either apatinib or APS
inhibited cell proliferation in a dose-dependent manner in ASPC-1 and PANC-1.
APS in combination with apatinib showed enhanced inhibitory effects on cell
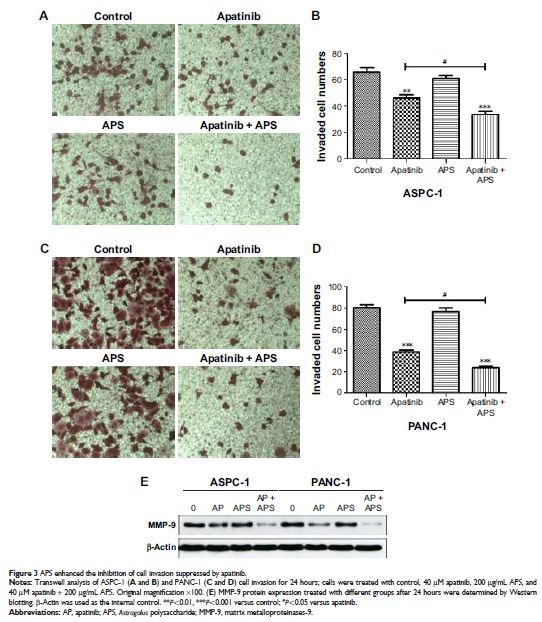
migration and invasion compared with apatinib monotherapy in ASPC-1 and PANC-1.
Meanwhile, APS combined with apatinib strongly increased cell apoptosis
percentage. Western blotting showed that the combination of APS and apatinib
significantly enhanced the downregulation of phosphorylated protein kinase B
(AKT) and extracellular signal-regulated kinase (ERK) (p-AKT and p-ERK) as well
as matrix metalloproteinases-9 (MMP-9) expression. In addition, both apatinib
and APS induced cellular autophagy. However, the expression of
autophagy-related proteins was not further elevated in the combination group.
Conclusion: The study first demonstrated that apatinib showed potentially
inhibitory effects in pancreatic cancer cells and that APS enhanced the
antitumor effects of apatinib through further downregulating the expression of
phosphorylation of AKT and ERK as well as MMP-9.
Keywords: AKT, ERK, digestive tumor, molecular mechanisms, molecular
targeted therapy, basic medical research, anti-angiogenesis drug