112158
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

DiGeorge 综合征关键区基因 9(一种长的非编码 RNA)在胃癌中的作用
Authors Ni C, Yang P, Guo J, Ye M
Received 11 January 2018
Accepted for publication 13 March 2018
Published 19 April 2018 Volume 2018:11 Pages 2259—2267
DOI https://doi.org/10.2147/OTT.S162253
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Justinn Cochran
Peer reviewer comments 2
Editor who approved publication: Dr XuYu Yang
Introduction: Long non-coding RNAs (lncRNAs) regulate and influence cancer cell
development and tumor formation. However, the role for lncRNAs in gastric
cancer has not been fully established. In this study, DGCR9 , a lncRNA, was significantly
upregulated in gastric cancer cell lines.
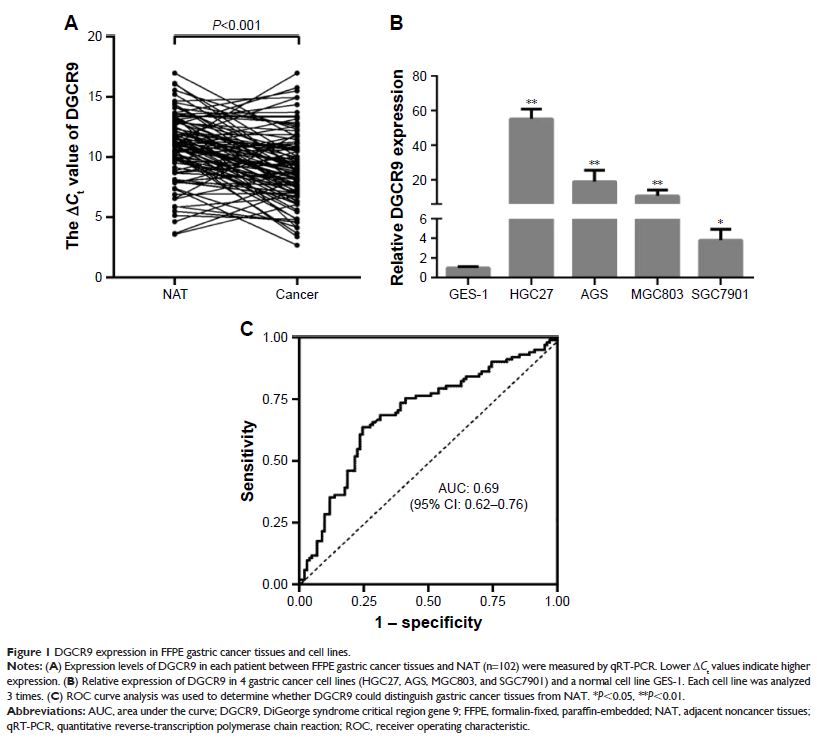
Methods: The expression levels of DGCR9 in each
patient between formalin-fixed, paraffin-embedded (FFPE) gastric cancer tissues
and adjacent noncancer tissues (NAT) (n=102) were measured by quantitative
reverse-transcription polymerase chain reaction (qRT-PCR). The effect of DGCR9 on cellular
proliferation, migration, and glucose uptake was investigated in vitro,
respectively.
Results: DGCR9 was shown to have increased expression in gastric cancer tissues and in
gastric cancer cell lines. Further, DGCR9 was
found to be associated with clinicopathological characteristics of patients
with gastric cancer. In particular, DGCR9 was
positively associated with lymph node invasion and tumor-node-metastasis (TNM)
stage in gastric cancer patients. By in vitro functional analysis, knockdown
of DGCR9 in gastric cancer cells
suppressed cellular proliferation, migration, and glucose uptake. In contrast,
overexpression of DGCR9 increased
each of these cancer cell characteristics.
Conclusions: DGCR9 was upregulated in
gastric cancer tissues and was shown to accelerate cellular proliferation,
migration, and glucose metabolism, all of which would promote the formation and
development of gastric cancer.
Keywords: long noncoding RNA, DGCR9, gastric cancer, proliferation,
migration, glucose metabolism