113568
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

对已接受重度治疗的转移性结直肠癌患者采用奥沙利铂治疗方案进行第三期或更晚期治疗的激发试验
Authors Yang Q, Huang YY, Jiang ZM, Wang HZ, Li WY, Zhang B, Xie DR
Received 16 October 2017
Accepted for publication 5 March 2018
Published 1 May 2018 Volume 2018:11 Pages 2467—2473
DOI https://doi.org/10.2147/OTT.S154220
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Andrew Yee
Peer reviewer comments 3
Editor who approved publication: Dr Ingrid Espinoza
Purpose: The third- or later-line therapy available often yield poor
survival benefit in patients metastatic colorectal cancer (mCRC). The
retrospective study aimed to evaluate efficacy of rechallenge of
oxaliplatin-containing regimens.
Patients and methods: Patients with mCRC who progressed from
fluoropyrimidine, oxaliplatin, and irinotecan in the first- and second-line
chemotherapy, were treated by reexposure to oxaliplatin-containing regimen.
Patients treated by anti-epidermal growth factor receptor (EGFR) antibodies
with irinotecan were included in the control arm.
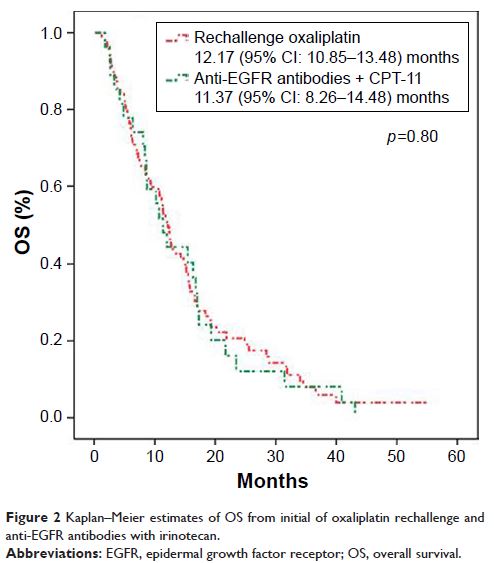
Results: Ninety-five and 29 patients were treated with
either oxaliplatin reexposure or anti-EGFR antibodies with irinotecan,
respectively, as the third- or later-line therapy. The median time to treatment
failure (TTF) and overall survival (OS) was 3.77 and 12.17 months in the
oxaliplatin arm, with 4.77 months of TTF and 11.37 months of OS in the control
arm; there was no significance between the 2 arms (p >0.05).
Oxaliplatin reexposure resulted in 6.3% objective response rate with no
complete response, 6 partial response, 39 stable disease, and 37 progressive
disease. The disease control rate was 47.4% (45/95). The multivariate analysis
found that patients who achieved disease control by oxaliplatin reexposure had
a superior TTF (6.13 vs 1.7 months, p <0.001) and OS
(15.73 vs 6.27 months, p <0.001)
compared with those presenting with progressive disease.
Conclusion: This study showed that rechallenge of
oxaliplatin-containing chemotherapy in the third- or later-line therapy may
lead to tumor control and improved survival in mCRC patients, which was
equivalent to that of anti-EGFR antibodies with irinotecan.
Clinical significance: Rechallenge of oxaliplatin-containing regimens
in the third- or later-line of therapy is a common practice, despite few
evidence available. The present study found that rechallenge of
oxaliplatin-containing regimens produced equivalent tumor control and survival
benefit to that of anti-EGFR antibodies with irinotecan in mCRC.
Keywords: rechallenge,
oxaliplatin, colorectal cancer, anti-epidermal growth factor receptor