113173
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

基于培美曲塞的治疗方案对晚期非小细胞肺癌患者表皮生长因子受体基因突变的疗效 一个系统的回顾
Authors Han BH, Yang LL, Wang X, Yao LD
Received 17 November 2017
Accepted for publication 10 February 2018
Published 12 April 2018 Volume 2018:11 Pages 2121—2129
DOI https://doi.org/10.2147/OTT.S157370
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Lucy Goodman
Peer reviewer comments 2
Editor who approved publication: Dr Carlos Vigil Gonzales
Abstract: Pemetrexed-based
chemotherapy regimens (pem regimens) are the standard first-line treatment
option in patients with non-squamous non–small cell lung cancer (NSCLC). The
objective of this systematic review was to assess the efficacy of pemetrexed in
the context of epidermal growth factor receptor (EGFR )
mutation-positive NSCLC following the failure of EGFR –tyrosine
kinase inhibitor (TKI) treatment. We searched biomedical literature databases
(PubMed, EMBASE, and the Cochrane library) and conference proceedings for
studies evaluating the efficacy of pemetrexed monotherapy or pemetrexed
combined with platinum or any other chemotherapeutic agent in EGFR –mutation-positive NSCLC
after EGFR -TKI failure. We extracted
data of primary outcomes of interest (progression-free survival [PFS], overall
survival [OS], and overall response rate [ORR]). The weighted median PFS, OS,
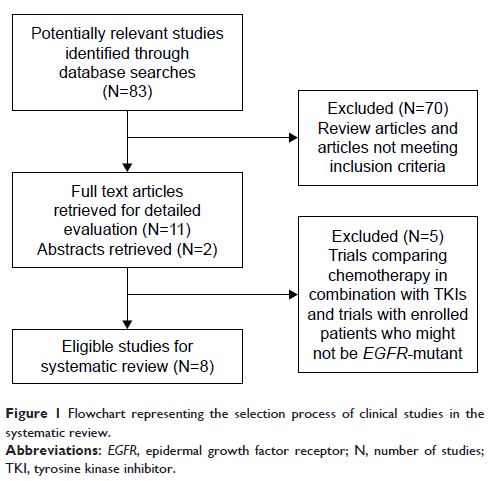
and ORR were then calculated. Of 83 potentially relevant studies, eight (three
randomized studies and five retrospective studies) were identified (involving
1,193 patients) and included in this systematic review, with 640 patients
receiving pem regimens. The weighted median PFS, median OS, and ORR for
patients treated with pem regimens were 5.09 months, 15.91 months,
and 30.19%, respectively. Our systematic review results showed a favorable
efficacy profile of pem regimens in NSCLC patients with EGFR mutation after EGFR –TKI failure.
Keywords: pemetrexed, advanced non–small cell lung cancer, epidermal growth
factor receptor, tyrosine kinase inhibitor