113173
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

The 95RGD97 sequence on the Aα chain of fibrinogen is essential for binding to its erythrocyte receptor
Authors Carvalho FA, Guedes AF, Duval C, Macrae FL, Swithenbank L, Farrell DH, Ariëns RAS, Santos NC
Received 19 October 2017
Accepted for publication 12 January 2018
Published 3 April 2018 Volume 2018:13 Pages 1985—1992
DOI https://doi.org/10.2147/IJN.S154523
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Govarthanan Muthusamy
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Thomas J Webster
Background: Erythrocyte
aggregation, a cardiovascular risk factor, is increased by high plasma
fibrinogen levels. Here, the effect of different fibrinogen mutations on
binding to its human erythrocyte receptor was assessed in order to identify the
interaction sites.
Methods: Three fibrinogen variants were tested, specifically
mutated in their putative integrin recognition sites on the Aα chain (mutants
D97E, D574E and D97E/D574E) and compared with wild-type fibrinogen.
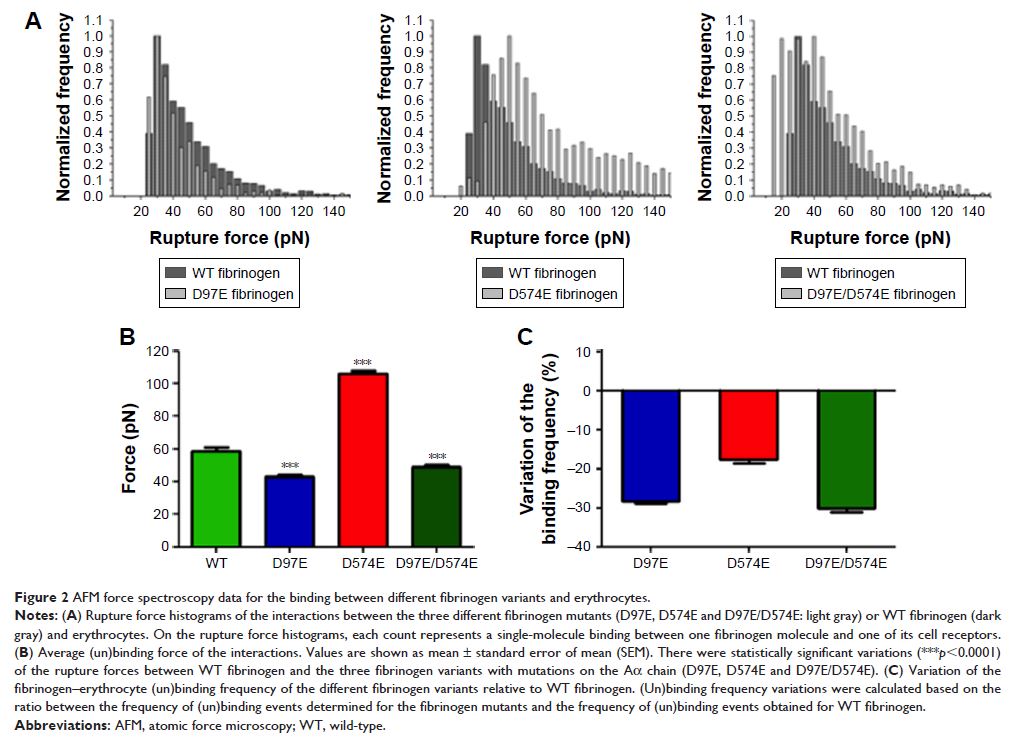
Results: Atomic force microscopy-based force spectroscopy
measurements showed a significant decrease both on the fibrinogen–erythrocyte
binding force and on its frequency for fibrinogen with the D97E mutation,
indicating that the corresponding arginine–glycine–aspartate sequence (residues
95–97) is involved in this interaction, and supporting that the fibrinogen
receptor on erythrocytes has a β3 subunit.
Changes in the fibrin clot network structure obtained with the D97E mutant were
observed by scanning electron microscopy.
Conclusion: These findings may lead to innovative perspectives on
the development of new therapeutic approaches to overcome the risks of fibrinogen-driven
erythrocyte hyperaggregation.
Keywords: atomic force
microscopy, fibrinogen, fibrin clot, erythrocyte aggregation, mutant protein