113173
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

多重治疗与阿帕替尼及拓扑替康相结合治疗铂类耐药复发卵巢癌患者: 3 个病例的报告
Authors Cheng Y, Zhang J, Geng HY, Qin SK, Hua HQ
Received 26 November 2017
Accepted for publication 22 February 2018
Published 6 April 2018 Volume 2018:11 Pages 1989—1995
DOI https://doi.org/10.2147/OTT.S158141
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Cristina Weinberg
Peer reviewer comments 2
Editor who approved publication: Dr XuYu Yang
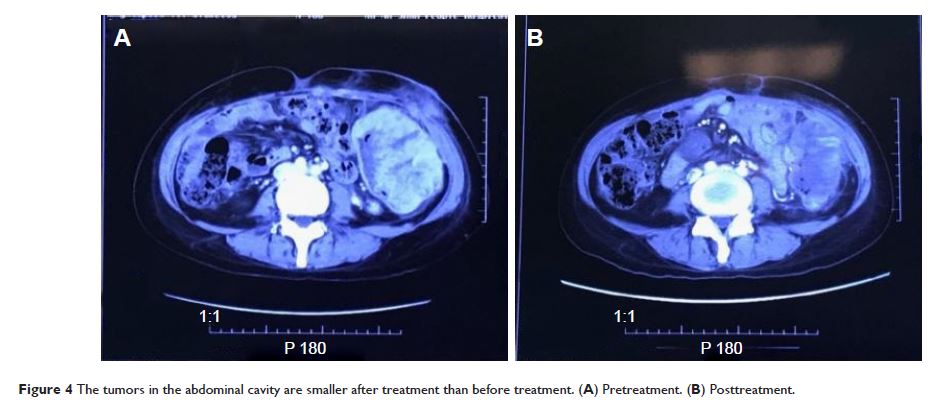
Abstract: The aim of this study was to
observe the efficacy and safety of apatinib combined with toptecan therapy in
the multiline treatment of platinum-resistant recurrent ovarian cancer
patients. The clinical records of three patients with platinum-resistant
recurrent ovarian cancer treated with apatinib combined with toptecan therapy
were analyzed and followed up for 3 months, and the related literatures were
reviewed. The three patients achieved partial response and the tumor marker
CA125 levels decreased significantly as an outcome of the treatment. Major
adverse reactions were hypertension, hand–foot skin reaction, and anemia, which
were manageable with medication. Apatinib combined with toptecan multiline
therapy in the treatment of platinum-resistant recurrent ovarian cancer
patients is effective, and the adverse effects are tolerated. Large-scale studies
should be conducted to further determine the efficacy and safety of this
treatment protocol.
Keywords: ovarian cancer,
apatinib, topotecan, gynecological oncology