113173
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

乙型肝炎血清核心抗体水平预测接受核苷(酸)类似物治疗的高病毒载量慢性乙型肝炎患者中的 HBeAg 血清转化
Authors Cai S, Li Z, Yu T, Xia M, Peng J
Received 18 January 2018
Accepted for publication 19 February 2018
Published 3 April 2018 Volume 2018:11 Pages 469—477
DOI https://doi.org/10.2147/IDR.S163038
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Cristina Weinberg
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Background: Patients with chronic hepatitis B virus (HBV) infection who are hepatitis
B virus e antigen (HBeAg) positive are increasingly being treated with
nucleos(t)ide analogs (NUCs). However, the predictive value of serum hepatitis
B virus core antibody (HBcAb) levels for HBeAg seroconversion among patients
with high viral load remains unclear.
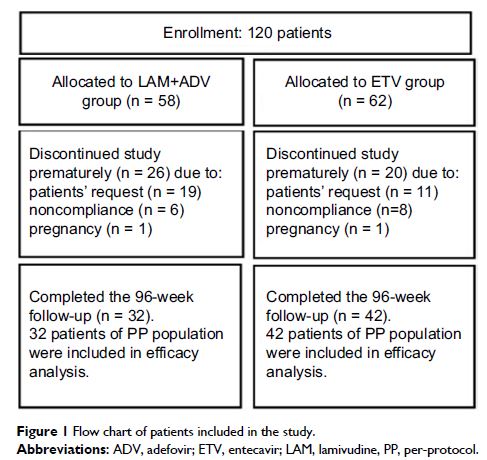
Methods: This study consisted of 74 patients with high viral load (HBV DNA >1
× 107 copies/mL)
enrolled in a multicenter, randomized, controlled trial, treated with
lamivudine and adefovir (N = 32) or entecavir (N = 42) for up to 96 weeks.
Serum HBV DNA, quantitative hepatitis B virus surface antigen (HBsAg), HBeAg,
and HBeAb was tested at each visit. Quantitative HBcAb evaluation was conducted
for all the available samples in the trial, by using a newly developed
double-sandwich anti-HBc immunoassay.
Results: Serum HBcAb levels were significantly higher in patients with a
serum alanine aminotransferase (ALT) level more than five times the upper limit
of normal (ULN) compared with patients with ALT levels within 5 × ULN (4.25 ±
0.61 vs. 3.94 ± 0.47 log10 IU/mL, P = 0.0345).
Patients with HBeAg seroconversion were associated with a higher level of HBcAb
at baseline, compared with those without HBeAg seroconversion (4.38 ± 0.54 vs.
4.02 ± 0.58 log10 IU/mL, P = 0.029). The area under
receiver operating characteristic curve of baseline HBcAb for HBeAg
seroconversion was 0.71 (95% CI: 0.55–0.86, P = 0.013).
When the baseline HBcAb level was >4.375 log10 IU/mL, the sensitivity and specificity to predict HBeAg
seroconversion at week 96 were 62.5% and 74.2%, and the positive likelihood
ratio (LR) and negative LR were 2.42 and 0.51, respectively. The multivariate
analysis result indicated that baseline serum HBcAb level was the only
independent predictor for HBeAg seroconversion at week 96, with an odds ratio
of 4.78.
Conclusion: Baseline serum HBcAb level >4.375 log10 IU/mL could satisfactorily predict HBeAg seroconversion among
patients with high viral load treated with NUC.
Keywords: chronic hepatitis B, HBV: hepatitis B core antibody, hepatitis B e
antigen, seroconversion, high viral load